
Lysosomes are the main degradation and recycling center organelles in eukaryotic cells. Our group mainly uses the popular animal model Drosophila to study the genetics, cell biology, biochemistry, and the physiological and pathological roles and mechanisms of lysosomal degradation pathways. More recently, we developed an interest into how all these routes are coordinated not only in Drosophila but also in cultured human cells. For example, we use cell biological and biochemical experiments in the lab to explore the dynamic nature of lysosomes in mammalian cell culture and Drosophila tissues, investigating how their lipid and protein compositions adapt to stress such as starvation, change during brain aging, and differ between neurons and glia.
Our recent projects primarily focus on three major lyosomal degradation routes: autophagy, endocytosis, and phagocytosis.
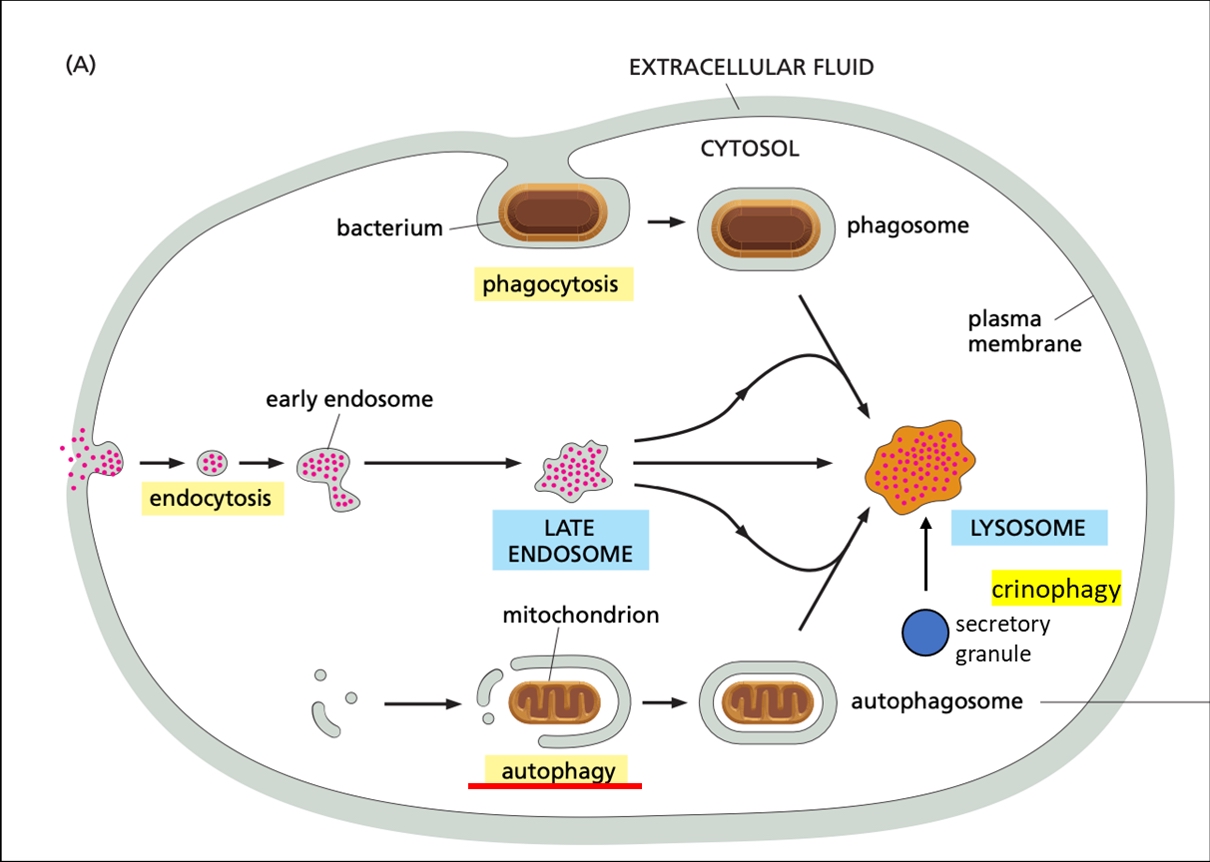
Figure 1. Main delivery routes of to-be-degraded cargo into lysosomes. Endocytosis takes up extracellular material and packages it into endosomes. Cargos are typically macromolecules and extracellular fluid, but also receptor proteins embedded into the plasma membrane. Phagocytosis is a type of endocytosis of particles > 0.5 μm that relies on cell surface receptors. Cargos range from bacteria, viruses, and apoptotic cell fragments to inert particles such as asbestos. Autophagy encapsulates intracellular components into a double-membrane transport vesicle (autophagosome), which then fuses with the lysosome. Cargos can be organelles, macromolecules and their assemblies, including more-or-less random portions of the cytoplasm. Crinophagy ensures the elimination of unwanted secretory granules via direct fusion with the lysosome.
Autophagy
Autophagy (cellular self-digestion by lysosomes) is observed in all eukaryotic cells. Autophagy is upregulated for example in response to starvation to ensure survival by recycling dispensable cellular constituents for re-use in synthetic processes. In animals, autophagy has been adopted to serve additional functions beyond starvation survival that include regulation of lifespan, stress responses, cellular homeostasis, cell death, immunity, cancer, neurodegeneration diseases, obesity and lipid metabolism, and more. The biomedical relevance of autophagy is well-established by now, and it is also evident from the 2016 Nobel Prize awarded to a yeast autophagy researcher. Selective autophagy enables the targeted recognition, sequestration, and removal of specific cellular components (including damaged or excess organelles, protein aggregates, and invading pathogens) through dedicated cargo recognition mechanisms. We have projects looking into the selective degradation of ubiquitinated proteins, ER (endoplasmic reticulum), and damaged lysosomes (that is, aggrephagy, ER-phagy, and lysophagy, respectively). We also focus on the fusion machinery between autophagosomes and lysosomes and how it is regulated by lipids and other cytoplasmic proteins. Such experiments involve the identification of interacting partners of selected components of the fusion machinery using both in vivo and in vitro approaches. Candidate interactions are reconstituted using purified components, and the functional impact of these interactions is subsequently examined through biochemical and genetic analyses.
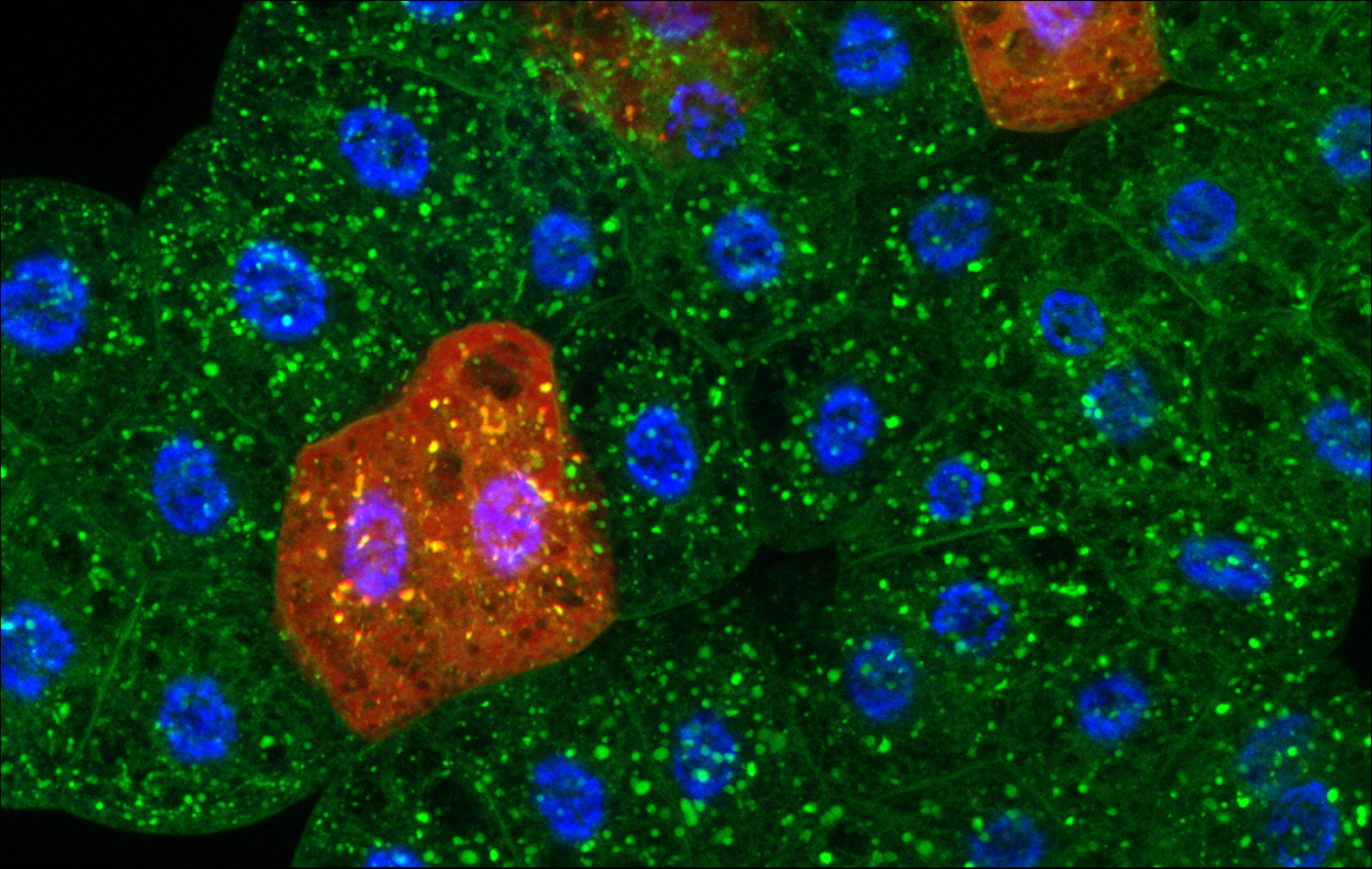
Figure 2. Starvation-induced ER-phagy assay using a mosaic genetic approach in the fat body of Drosophila. This organ is functionally analogous to the mammalian liver and fat and it exhibits robust autophagy in response to starvation stress. Therefore, multiple forms of autophagy can be effectively monitored in this tissue, including ER-phagy, which mediates the selective degradation of the endoplasmic reticulum (ER). In the image, Sec61-GFP (green) labels the ER, while cells shown in red indicate clones in which one of the ER-phagy receptor genes has been silenced.
Endocytosis
Taking up material with endocytosis is a prerequisite of normal cell function. Alterations in endosomal trafficking can lead to severe defects in cellular and organismal physiology. Transport of endocytic cargo in the endosomal network of eukaryotic cells involves directed movement, maturation, and ultimately the fusion of carrier vesicles with lysosomes. Several proteins and multiprotein complexes are required for propelling the steps of endosomal maturation. Our investigations aim to understand the function of previously uncharacterized proteins with presumable endocytic functions using the highly endocytic larval nephrocytes of the fruit fly as an experimental model system.
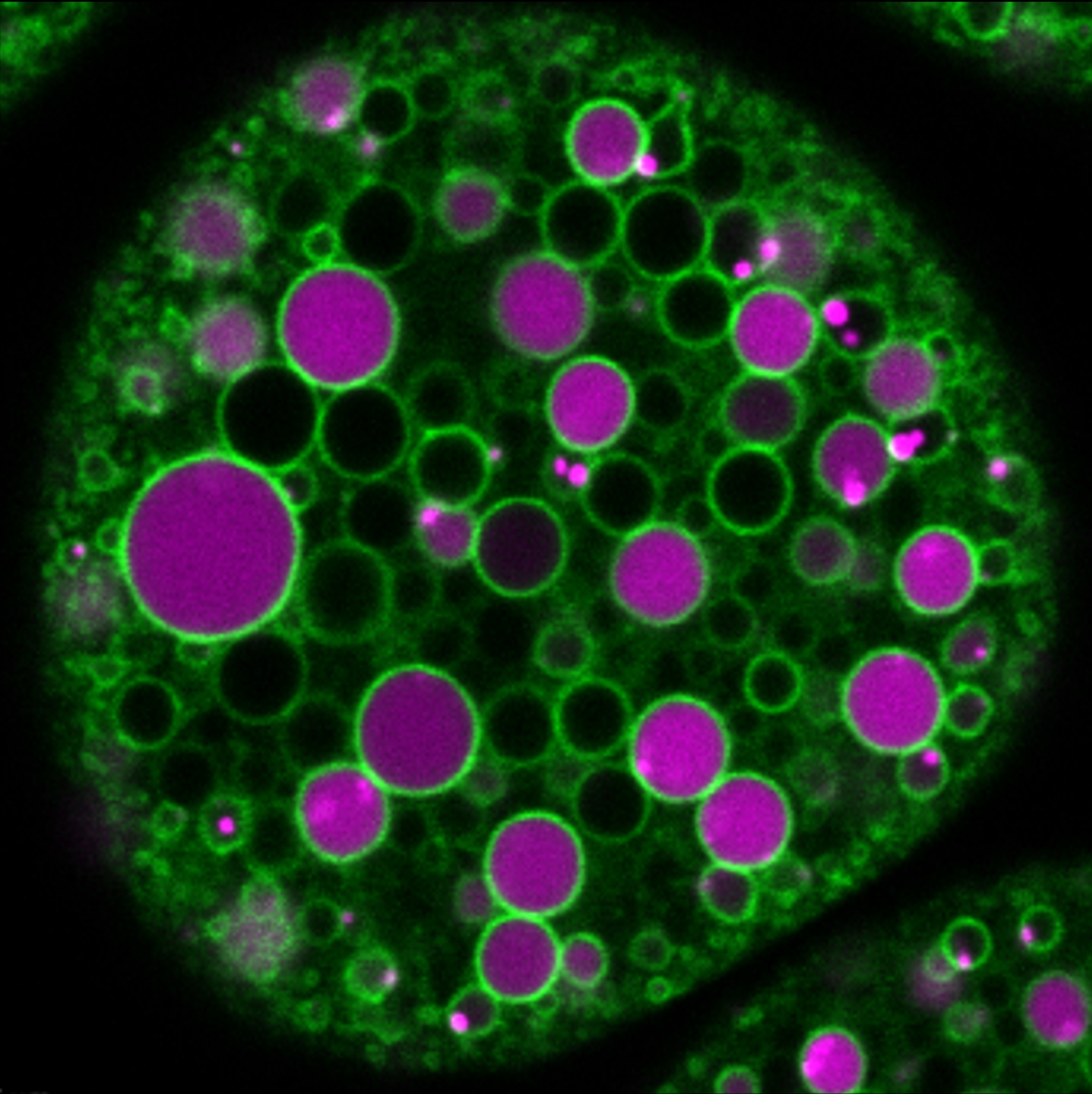
Figure 3. Confocal microscopic image of an ex vivo cultured living nephrocyte containing a fluorescently labelled endocytic tracer (magenta) and a fluorescently tagged endosomal protein (green). The tracer is transported to spherical endosomes after it was taken up by the cell via endocytosis from the culture media. Green „rings” represent endocytic vesicles with the tagged protein recruited to the outer leaflet of their limiting membrane. Experimental setups like this enable the investigation of endocytic trafficking through the simultaneous visualization of the endocytic cargo and regulator proteins of the endosomal maturation process.
Phagocytosis
The process of phagocytosis entails the uptake of particles from the extracellular space, which normally reach the lysosomes and are degraded. Professional phagocytes, such as macrophages, are equipped with a battery of receptors, adhesion proteins, and their cytoskeleton is very dynamic, all of which are important for their phagocytic ability. We study phagocytosis in two different cell types in Drosophila. First, our macrophage studies focus on understanding the mechanisms and importance of the uptake of extracellular matrix fragments, and the engulfment of apoptotic debris (efferocytosis). We aim to identify general mechanistic components, as well as factors specific to a given subtype of phagocytosis. Second, we study glia, which are the only phagocytes in the nervous system in physiological conditions. We recently found that glia use a special pathway called LC3-associated phagocytosis to clear cellular debris after injury. We now investigate the role of this process in the correct wiring of the fly visual system via synapse pruning. We also study how glia fight neurodegenerative diseases such as Alzheimer’s disease via enhanced degradation of toxic proteins.
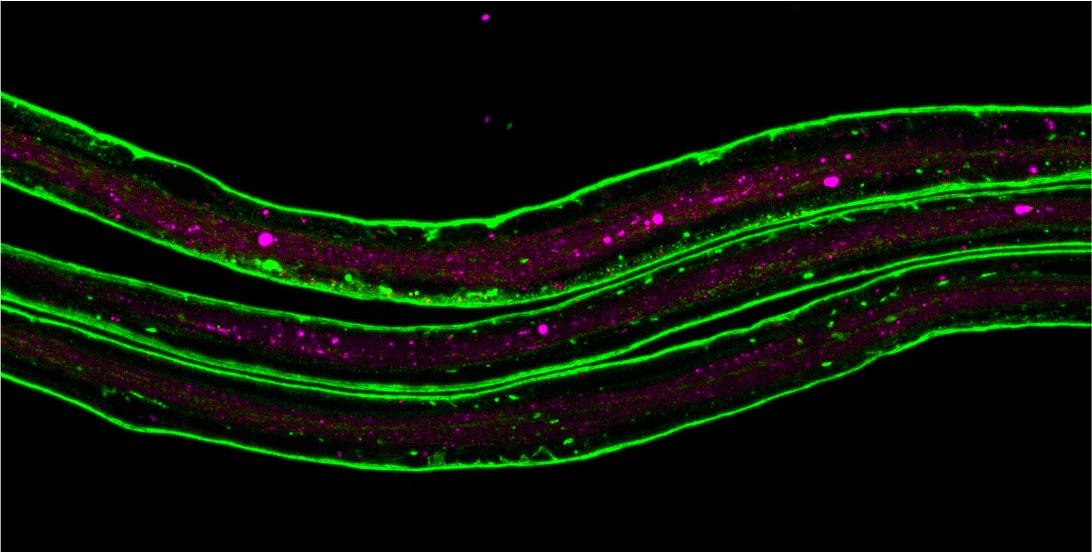
Figure 4. The image shows larval abdominal nerves, where the extracellular matrix protein Laminin B1 is labeled in green, and glial lysosomes are labeled in magenta. We use this system to uncover how glia cells reshape their ECM environment during normal development and following injury by clearing excess matrix material. Aided by the powerful genetic tools available in Drosophila, we constructed screening systems to identify key factors in matrix uptake, which we can further investigate to understand the mechanisms underlying phagocyte-mediated engulfment of ECM fragments.
Selected papers
Selective autophagy fine-tunes Stat92E activity by degrading Su(var)2-10/PIAS in Drosophila glia. Vincze V, Esküdt Z, Fehér-Juhász E, Chhatre AS, Jipa A, Galambos AR, Feil-Börcsök D, Bence M, Juhász G, Szabó Á. Life Sci Alliance. 2026 Jan 7;9(3):e202503375. doi: 10.26508/lsa.202503375.
Lysosomal activity depends on TRPML1-mediated Ca2+ release coupled to incoming vesicle fusions. Bhattacharjee A, Abuammar H, Juhász G. J Biol Chem. 2024 Dec;300(12):107911. doi: 10.1016/j.jbc.2024.107911
PtdIns4P is required for the autophagosomal recruitment of STX17 (syntaxin 17) to promote lysosomal fusion. Laczkó-Dobos H, Bhattacharjee A, Maddali AK, Kincses A, Abuammar H, Sebők-Nagy K, Páli T, Dér A, Hegedűs T, Csordás G, Juhász G. Autophagy. 2024 Jul;20(7):1639-1650. doi: 10.1080/15548627.2024.2322493
LC3-associated phagocytosis promotes glial degradation of axon debris after injury in Drosophila models. Szabó Á, Vincze V, Chhatre AS, Jipa A, Bognár S, Varga KE, Banik P, Harmatos-Ürmösi A, Neukomm LJ, Juhász G. Nat Commun. 2023 May 29;14(1):3077. doi: 10.1038/s41467-023-38755-4.
Analysis of Drosophila Atg8 proteins reveals multiple lipidation-independent roles. Jipa A, Vedelek V, Merényi Z, Ürmösi A, Takáts S, Kovács AL, Horváth GV, Sinka R, Juhász G. Autophagy. 2021 Sep;17(9):2565-2575. doi: 10.1080/15548627.2020
Vps8 overexpression inhibits HOPS-dependent trafficking routes by outcompeting Vps41/Lt. Lőrincz P, Kenéz LA, Tóth S, Kiss V, Varga Á, Csizmadia T, Simon-Vecsei Z, Juhász G. Elife. 2019 Jun 13;8:e45631. doi: 10.7554/eLife.45631.
Molecular mechanisms of developmentally programmed crinophagy in Drosophila. Csizmadia T, Lőrincz P, Hegedűs K, Széplaki S, Lőw P, Juhász G. J Cell Biol. 2018 Jan 2;217(1):361-374. doi: 10.1083/jcb.201702145.
Microenvironmental autophagy promotes tumour growth. Katheder NS, Khezri R, O'Farrell F, Schultz SW, Jain A, Rahman MM, Schink KO, Theodossiou TA, Johansen T, Juhász G, Bilder D, Brech A, Stenmark H, Rusten TE. Nature. 2017 Jan 19;541(7637):417-420. doi: 10.1038/nature20815
Rab2 promotes autophagic and endocytic lysosomal degradation. Lőrincz P, Tóth S, Benkő P, Lakatos Z, Boda A, Glatz G, Zobel M, Bisi S, Hegedűs K, Takáts S, Scita G, Juhász G. J Cell Biol. 2017 Jul 3;216(7):1937-1947. doi: 10.1083/jcb.201611027.
Autophagosomal Syntaxin17-dependent lysosomal degradation maintains neuronal function in Drosophila. Takáts S, Nagy P, Varga Á, Pircs K, Kárpáti M, Varga K, Kovács AL, Hegedűs K, Juhász G. J Cell Biol. 2013 May 13;201(4):531-9. doi: 10.1083/jcb.201211160.

scientific adviser

senior research associate

senior research associate

research associate

research associate

research associate

research associate

research associate

research associate

research associate

junior research associate

PhD student

PhD student

PhD student

PhD student

laboratory assistant

administrator expert

Szent-Györgyi student
 Gábor, JUHÁSZ
Gábor, JUHÁSZ
|
scientific adviser | publications | CV |
 Áron, SZABÓ
Áron, SZABÓ
|
senior research associate | publications | CV |
 Gábor, CSORDÁS
Gábor, CSORDÁS
|
senior research associate | publications | CV |
 András, BLASTYÁK
András, BLASTYÁK
|
research associate | publications | CV |
 Judit, BÍRÓ
Judit, BÍRÓ
|
research associate | publications | CV |
 Adél, HARMATOS-ÜRMÖSI
Adél, HARMATOS-ÜRMÖSI
|
research associate | publications | CV |
 Hajnalka, HEVÉRNÉ LACZKÓ-DOBOS
Hajnalka, HEVÉRNÉ LACZKÓ-DOBOS
|
research associate | publications | CV |
 András, JIPA
András, JIPA
|
research associate | publications | CV |
 Tamás, MARUZS
Tamás, MARUZS
|
research associate | publications | CV |
 Amruta NAIK
Amruta NAIK
|
research associate | publications | CV |
 Daniela EVDOKIMOVA
Daniela EVDOKIMOVA
|
junior research associate | publications | CV |
 Aishwarya, CHHATRE
Aishwarya, CHHATRE
|
PhD student | publications | CV |
 Virág, VINCZE
Virág, VINCZE
|
PhD student | publications | CV |
 Lilla, PÉTER
Lilla, PÉTER
|
PhD student | publications | CV |
 Dániel, DÉNES
Dániel, DÉNES
|
PhD student | publications | CV |
 Szilvia, BOZSÓ
Szilvia, BOZSÓ
|
laboratory assistant | ||
 Erzsébet, FEHÉRNÉ JUHÁSZ
Erzsébet, FEHÉRNÉ JUHÁSZ
|
administrator expert | publications | |
 Zsombor, ESKÜDT
Zsombor, ESKÜDT
|
Szent-Györgyi student |